Publications & press
News & updates
Oxylanthanum Carbonate Achieves Serum Phosphate Control with Significantly Lower Pill Burden in Dialysis Patients with Hyperphosphatemia

A Phase 2 Clinical Trial of Oxylanthanum Carbonate in Patients Receiving Maintenance Hemodialysis with Hyperphosphatemia – CJASN Manuscript – July 2025

Video: Interview with Dr. Glenn Chertow

AJN: High Phosphate-Binding Capacity of oxylanthanum carbonate with a Low Medication Volume: Comparison with Commercially Available Phosphate Binders

Video: Our Lead Asset (Oxylanthanum Carbonate)

Two-Way Randomized Crossover Study to Establish Pharmacodynamic Bioequivalence Between oxylanthanum carbonate and Lanthanum Carbonate

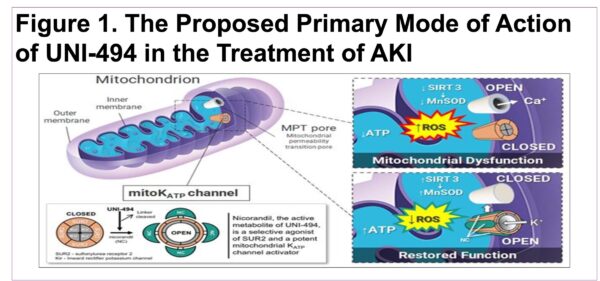
EC Pharmacology and Toxicology: Evaluation of UNI-494 in Acute Kidney Injury Treatment Efficacy When Administered After Ischemia-Reperfusion in a Rat Model

Pharmaceutical Chemistry Journal: Systemic Absorption of Oxylanthanum Carbonate is Minimal in Preclinical Models
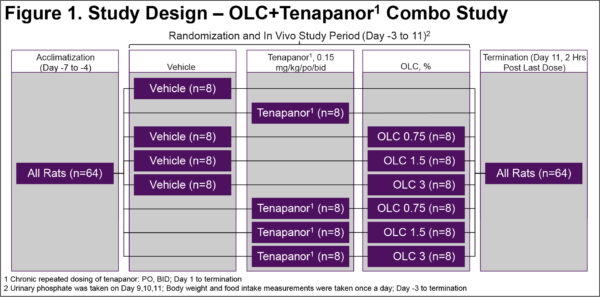
Combination Oxylanthanum Carbonate and Tenapanor Synergistically Lowers Urinary Phosphate Excretion in Rats
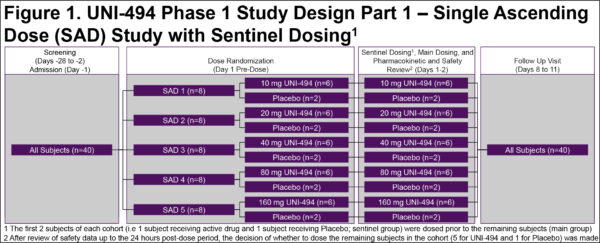
UNI-494 Phase I Safety, Tolerability, and Pharmacokinetics
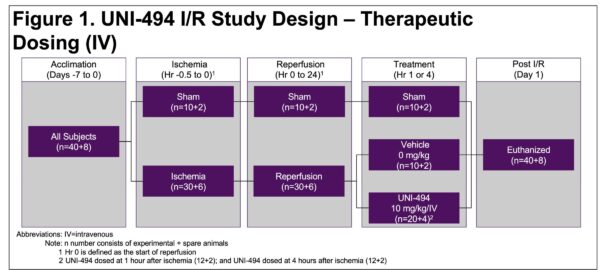
Intravenous UNI-494 Slows the Progression or Halts/Reverses Acute Kidney Injury When Administered After Ischemia/Reperfusion in Rats
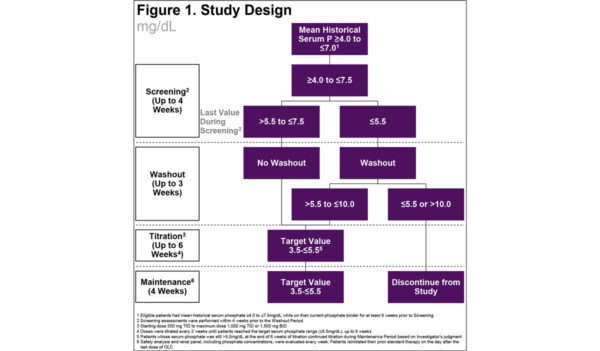
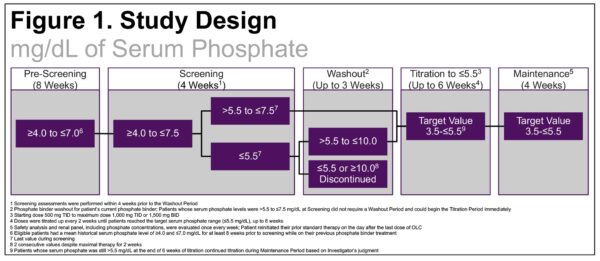
Effects of Oxylanthanum Carbonate in Patients Receiving Maintenance Hemodialysis with Hyperphosphatemia
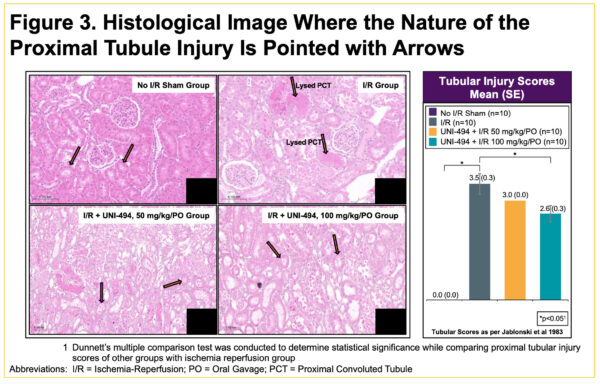
Oral Administration of UNI-494 Ameliorates Acute Kidney Injury in a Rat Model of Delayed Graft Function
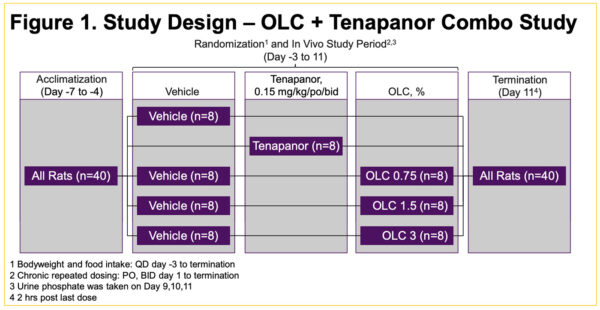
Enhanced Urinary Phosphorous Reduction: Comparative Study of Oxylanthanum Carbonate and Tenapanor in Rats
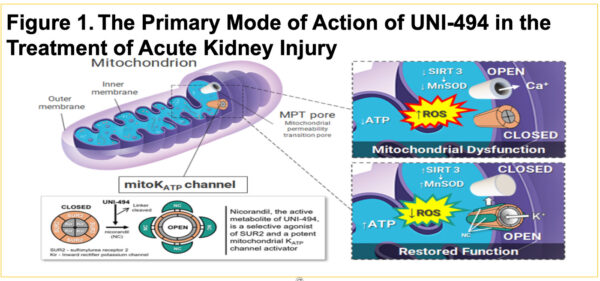
UNI-494 Phase I Safety, Tolerability and Pharmacokinetics: Trial in Progress
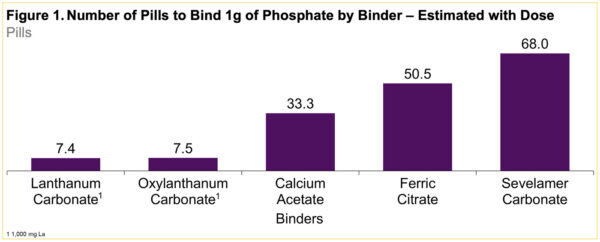
Oxylanthanum Carbonate for Hyperphosphatemia in ESKD: Tolerability Trial in Progress
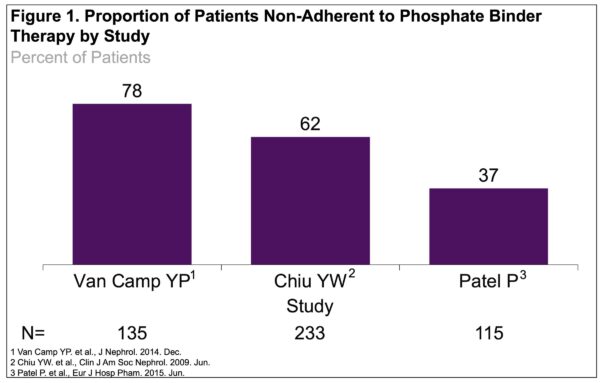
Renal Dietitians Perceive Phosphate Binder and Low Phosphorus Diet Non-Compliance as Top Reasons for Above Target Serum Phosphorus Concentrations
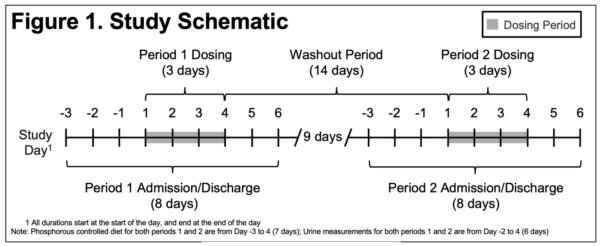
Two-Way Crossover Study to Establish Pharmacodynamic Bioequivalence Between OxylanthanumCarbonate and Lanthanum Carbonate

Video: About Unicycive